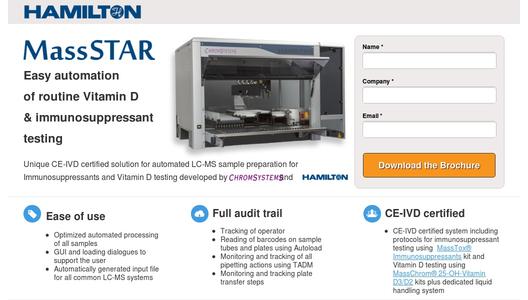
Chromsystems and Hamilton Robotics today announced the release of a complete CE-IVD marked solution that allows automated sample preparation for LC-MS/MS based clinical diagnostics.
The ready-to-use CE-IVD solution consists of MassSTAR which automates the sample preparation procedure and two Chromsystems’ CE-IVD certified kits, the MassChrom® 25-OH-Vitamin D3/D2 for Vitamin D profiling and the MassTox® Immunosuppressants for Therapeutic Drug Monitoring of Cyclosporine A, Tacrolimus, Sirolimus and Everolimus. The jointly developed MassSTAR enables laboratories to fully automate sample preparation for the testing of Vitamin D from serum and plasma as well as of Immunosuppressants from whole blood.
MassSTAR is based on the Microlab STAR line of instruments and was co-developed by Chromsystems and Hamilton as a ready to use and fully CE-IVD compliant workflow solution. The dedicated Graphical User Interface (GUI) and dialogues make it easy to use. Input worklists can be generated for all commonly used LC-MS/MS systems such as AB Sciex, Agilent, Shimadzu, Thermo and Waters. Special attention was paid to ensure full traceability of all samples and actions. During loading, barcodes of all samples and plates are read and traced. All actions on the instrument, such as pipetting and transport actions, are monitored as well. Hamilton’s Total Aspiration and Dispense Monitoring (TADM) technology is employed to tightly monitor pipetting of whole blood and exclude faulty samples.
"The increased use of LC-MS/MS as a gold standard for many parameters in diagnostics has led to an increased demand for automated CE-IVD validated solutions for sample preparation. This is true in particular for laboratories with a larger number of samples, where manual sample preparation can become a bottle neck. Partnering with Hamilton to automate our MassChrom ® 25-OH-Vitamin D3/D2 and MassTox® Immunosuppressants kits on the Microlab STAR line instruments has enabled the development of a unique CE-IVD validated workflow solution for two very common tests and exemplifies our commitment to provide innovative solutions for routine clinical diagnostics by mass spectrometry" says Michael Meier, General Manager at Chromsystems.
"At Hamilton we have a strong focus on, and expertise in, providing scientists with solutions that exactly fit with a lab's needs, but we also offer standardized solutions for a wide range of applications. We are very happy to work with Chromsystems as this partnership broadens our diagnostics portfolio with a unique pre-validated solution for LC-MS/MS based diagnostic purposes. Through Chromsystems' established knowledge in the diagnostic area, we can provide these combined solutions to diagnostic laboratories worldwide" says Dr. Mario Arangio, Director Standard Solutions at Hamilton Robotics.
For further information on the MassSTAR automated sample preparation system, please visit www.massstar.info/automation/.
About Chromsystems
Chromsystems is a leading global company providing ready-to-use reagent kits and supplies for routine clinical diagnostics by HPLC and LC-MS/MS, the latter representing the gold standard for a large number of parameters. Chromsystem’s product portfolio includes complete kits as well as quality controls and calibrators and enable any laboratory to introduce HPLC and LC-MS/MS methods into their diagnostic routine without prior technical expertise. The LC-MS/MS kits are comprehensively validated on all widely used tandem mass spectrometers. and are CE-IVD compliant, satisfying regulatory requirements for the laboratory. These high quality products are combined with an excellent support program and service for customers.
For more information visit http://www.chromsystems.com.
For more information, visit http://www.hamiltonrobotics.com.
* This product is not available in the USA